Overview
The ASPREE-XT Longitudinal Data Set (XT04) contains analytical data collected for the parent ASPREE Clinical Trial (2010 – June 2017), the Bridge period (June 2017 to 31 January 2018) and the ASPREE-XT cohort study (1 February 2018 onward) through to the participant’s fourth ASPREE-XT annual visit date (XT04), i.e., the participant’s fourth annual visit after 1 February 2018. For participants who did not have a fourth ASPREE-XT annual visit, the scheduled date of their fourth annual visit was used as a cut-off date for data capture. Milestone visits conducted after cessation of study medication, which were not included in the ASPREE Longitudinal Data Set (Version 3), have been included in this data set.
If an ASPREE participant withdrew prior to the start of ASPREE-XT, fact of death was included in this data set if the participant died before the date of the most recent linkage with the NDI. For Australia, this linkage was 8 January 2022 and for the US this was 2 November 2017.
NOTE: Sub-study data is not included in this data set. For more information on ASPREE sub-studies please see here.
ASPREE-XT Longitudinal Data Set (XT04) Structure
The ASPREE-XT Longitudinal Data Set (XT04) consists of nine sections (A-I) as seen in Table 1 below and on the panel to the left. This structure has been maintained from the ASPREE Longitudinal Data Set (Version 3) which was released in 2019 and the ASPREE-XT Longitudinal Data Set (XT02) which was released in 2021. The data set includes only structured data. For adjudicated endpoints, this means the data set includes the timing of the event (expressed in ‘days since randomisation’), the adjudicated event type and adjudicated sub-type. Unstructured data (such as PDF medical records) are not included.
Table 1. Structure of the ASPREE-XT Longitudinal Data Set (XT04).
| Section | Details |
|---|---|
| Section A | A.1 Baseline participant demographics |
| A.2 Longitudinal alcohol and smoking information | |
| Section B | B.1 Baseline past medical and cancer history |
| B.2 Longitudinal physical examination examination | |
| B.3 Longitudinal cancer screening | |
| B.4 Longitudinal pathology | |
| B.5 Longitudinal concomitant medication use | |
| B.6 Longitudinal family medical history | |
| B.7 Non-prescription medication use (Milestone visit only) | |
| Section C | C.1 Longitudinal 3MS examination |
| C.2 Longitudinal COWAT, HVLT-R, SDMT and Color Trails | |
| Section D | D.1 Longitudinal grip strength and gait speed |
| Section E | E.1 Longitudinal Life Disability |
| E.2 Longitudinal CES-D 10 | |
| E.3 Longitudinal SF-12 | |
| Section F | F.1 Primary and secondary endpoint data from 2018 New England Journal of Medicine papers |
| F.2 All reported endpoints | |
| F.3 Adjudicated secondary endpoints | |
| F.4 Hospitalisation for reasons other than endpoints | |
| F.5 Derived endpoints | |
| Section G | G.1 Study medication allocation and exposure |
| G.2 Study and prescription medication (Milestone visit only) | |
| G.3 Longitudinal aspirin use (XT) | |
| Section H | H.1 Visit completion |
| Section I | I.1 Baseline derived variables and exposure |
| I.2 Supplementary endpoint data (select adjudicated secondary endpoints only) | |
To see more information about each section, please click on the section of interest in the panel on the left.
Quality Control
ASPREE Web Accessible Relational Database (AWARD)
The ASPREE Web Accessible Relational Database (AWARD)-Data module supported data entry of study measures and events, participant booking, communication between study staff and GPs, study medication tracking and upload of supporting documentation for events. Each structured data field was subject to pre-programmed value ranges, process prompts and protocol compliance checks at the point of data entry, with the aim of preventing transcription errors in real time. Staff were alerted to any out-of-range or missing values when data were saved, and prompted to double check that these data were correct. For more information on the AWARD suite please see the Data Collection section.
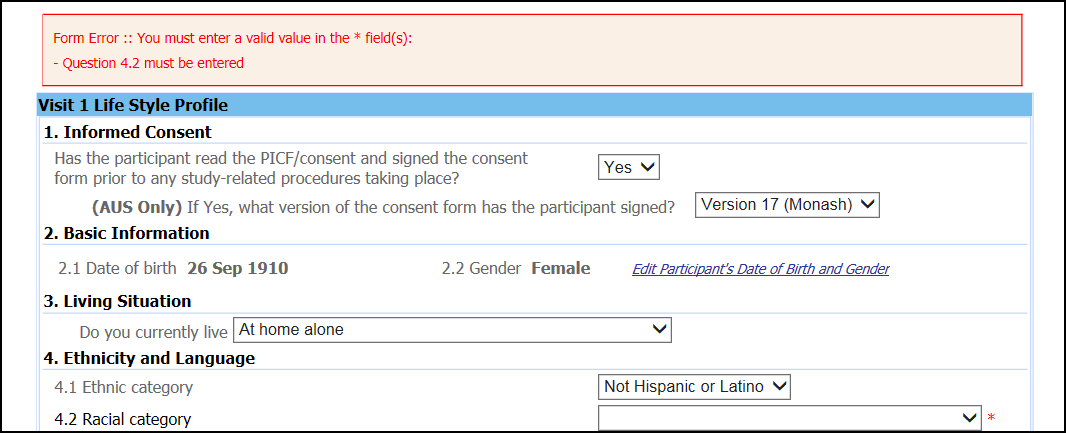
In addition to pop-up warnings, page submission restrictions were introduced. Each electronic Case Report Form (eCRF) included a ‘save draft’ and ‘submit’ button. The ‘save draft’ allowed staff to enter pieces of data without restrictions. However, when the ‘submit’ option was utilised the completeness of data entry was confirmed by the web-portal according to pre-specified validation rules. If a mandatory field was missed, a red error message appeared to alert staff (see Figure 2).
Throughout longitudinal data collection, eCRFs with a status of ‘Draft’ or ‘Not Entered’ appeared on a list of visits with missing data. This list was available through the web portal to all staff and was supervised by site managers. Visits were removed from the list only when all data was entered.

Wherever possible, staff entered raw data via the web application (e.g. individual blood pressure readings) and AWARD was programmed to calculate additional variables from the raw data (e.g. mean blood pressure). To aid the timely completion of study activity, AWARD also supported complex operational tasks such as visit bookings.
During ASPREE-XT, clinical event data were manually abstracted from clinical records. To minimise the need for interpretation by data collectors, clinical event record forms prompted staff to transcribe key elements that comprised the event definition, rather than to interpret clinical information and record outcomes. Logic checks, particularly related to illogical dates, were implemented to screen for transcription errors. To ensure correctness, primary and secondary endpoints were adjudicated by at least two clinical experts based on raw transcribed data and PDF copies of clinical records.
Randomisation Restrictions
Protocol deviations were minimised through the introduction of web-based randomisation restrictions. Staff were unable to randomise participants with ineligible data, for example low hemoglobin, abnormal blood pressure, history of bleeding ulcer or ineligible date of birth.
Data Queries
All analytical data in the ASPREE-XT Longitudinal Data Set (XT04) was subject to missing data queries. Numerical data was subject to out-of-range value queries and out-of-range change over time queries. The upper and lower limits for value queries were determined by the ASPREE International Data Management Committee and can be found in the ASPREE-XT Longitudinal Data Set (XT04) Data Dictionary. The mean change between annual visit values were calculated for numerical values. A change in data that was more than two standard deviations from the mean change was considered an out-of-range change over time and was flagged for review.
AWARD-Data included a tool for real-time querying of missing, out-of-range values and out-of-range change over time data. This tool was known as the staff ‘Action List’. The Action List was displayed on the home page of AWARD-Data to ensure that staff were aware of any queries linked with their user account.
When out-of-range values and data that represented an out-of-range change was entered, a query item was automatically added to the Action List of the appropriate staff member. Similarly, if eCRFs were saved with missing data, a query item was automatically added to the Action List of the appropriate staff member.
Staff were asked to respond to each query by reviewing the source documentation and either fixing the data (by entering missing data or correcting transcription errors) or by entering a response on the Action List confirming that the data was correct.
Data that were confirmed to be correct according to source documentation but considered to be unlikely or improbable, were reviewed and adjudicated by the IDMC. Data adjudicated as implausible (outside the possible range for humans) were removed and considered to be missing due to staff error at the time of data collection.
Commentary Values
Interpretation of ASPREE-XT data cannot be undertaken without consideration of the data collection and data querying processes. To ensure that the data is analysed in the context intended by the investigators, all data fields have been annotated with a commentary code.
The commentary codes provide explanation with regard to missing data, out of range data, alterations to data collection methods and other special considerations.
Staff Training and Monitoring
All staff were required to attend an initial training session covering good research practice, clinical research ethics and relevant Standard Operating Procedures (SOPs) that described all study procedures.
Visit Monitoring
Each staff member involved in visit conduct activity had two to four visits (minimum of two of each visit type) assessed each year by a designated monitor. Monitors assessed staff for their adherence to protocol and compliance with visit conduct and test administration SOPs by reviewing areas such as adherence to compulsory scripts, following predetermined test administration sequences and timings, collection of endpoint related information, and accuracy of questionnaire administrations. Assessment scoring was also reviewed and confirmed.
File Monitoring
A designated file monitor reviewed data collection processes by checking the source documents for five participant files per staff member every year. Source documents were assessed for completeness (including both data and ‘office use’ fields) and for transcription accuracy by comparing corresponding entries on eCRFs on the web portal.
Online Monitoring
As discussed above, AWARD-Data was designed to internally monitor many aspects of data entry.
Site Monitoring
All sites were monitored annually by trained clinical research site monitors following GCP guidelines. Sites were assessed for adherence to standardised operating procedures for drug log management, report production, ethics approvals, regulatory documents, work practices, equipment management and communication.